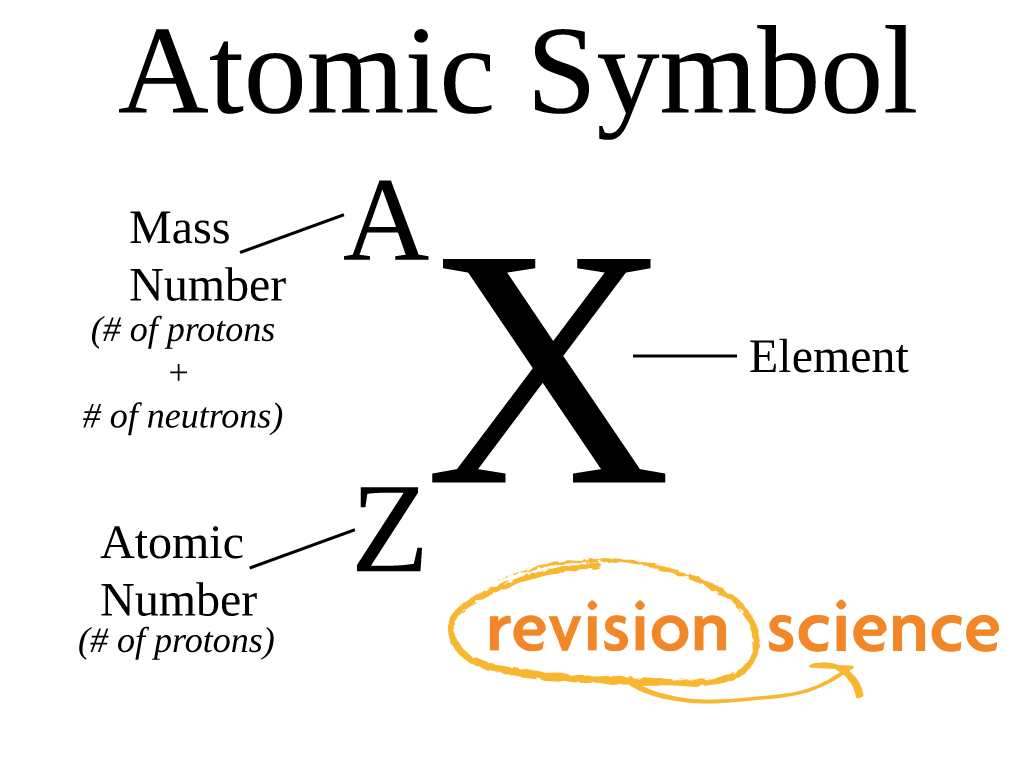
A chemical reaction is a rearrangement of atoms. Compounds are formed by the combination of two or more atoms.All atoms of a given element are identical.All matters are made out of atoms and atoms can’t be broken down further.Sometimes, there are very weak attractions between atoms.Ītom’s structure was determined by a series of experiments carried out by various scientists. According to their electron donating or withdrawing abilities, they can form covalent bonds or ionic bonds. All elements have a diatomic or polyatomic arrangement to become stable except the Nobel gases. Atoms can join with other atoms in various ways, thus form thousands of molecules. Same type of atoms may differ due to the number of neutrons present, and these are known as isotopes. The attractive forces between the positive charged nucleus (positive charge due to protons) and the negatively charged electrons maintain the atom’s shape.Ītoms of the same kind have similar protons and electrons. Other than neutrons and protons there are other small sub atomic particles in the nucleus, and there are electrons circling around the nucleus in orbitals. Atom is made up of a nucleus, which has protons and neutrons. Normally atoms are in the Angstrom range. They are so tiny that we can’t even observe with our naked eye. When this happens, the properties of single elements vary and give rise to novel mixtures.Ītoms are the small building blocks of all existing substances. They form various combinations between them or with other elements in order to exist. The single elements are hardly stable under natural conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
